 The H 2 S molecule possesses a mutual bond angle of 92.1° between the bonded atoms. H 2 S does not possess any hybridization as per Drago’s rule. Each fluorine takes three lone pairs, so there are 34 (8 + 4×6) 2 electrons left which go to the sulfur as a lone pair: The central atom has 4 atoms connected to it, and one lone. The ideal electron pair geometry of H 2 S is tetrahedral. Sulfur is the central atom: There are 6 + 4×7 34 valence electrons, and 8 are taken for making 4 covalent bonds. Hence, the molecular shape or geometry for SiCl4 is tetrahedral. The molecular geometry or shape of H 2 S is bent, angular or V-shaped. 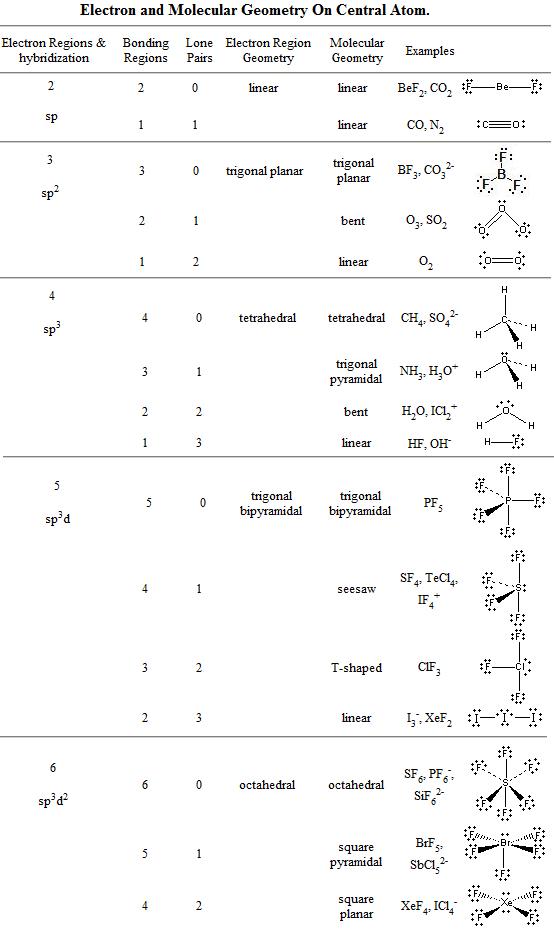
Specifically, VSEPR models look at the bonding and molecular geometry of organic molecules and polyatomic ions. So, that bends the axial fluorines together a bit.\): Geometric structure of methyl isocyanate (CH 3–N=C=O), note there is no rotation around the double bonds only the single CN bond can rotate. So, as per the VSEPR chart, if the central atom of a molecule contains 0 lone pairs and is cornered by four surrounding atoms, then the molecular shape of that molecule is tetrahedral in nature. The valence shell electron pair repulsion model is often abbreviated as VSEPR (pronounced " vesper" ) and is a model to predict the geometry of molecules. Note though that the structure is distorted a bit due to the repulsive forces of the lone pair of electrons you see (not bonded). With one lone pair of valence electrons, you get a seesaw molecular geometry. 
These are arranged in a trigonal bipyramidal shape with 102 F-S-F bond angles between the equatorial fluorine atoms and 173 between the axial fluorine atoms. Figure 6.3.2 Geometries for Species with Two to Six Electron Groups. Sulfur tetrafluoride has 5 regions of electron density around the central sulfur atom (4 bonds and one lone pair). 
Therefore, you can put 6x4 on each fluorine, 2x4 to account for four single bonds, and 2 for the last 2 valence electrons available.Īs a result, you have 5 electron groups, so the electron geometry would be trigonal bipyramidal. The VSEPR model explains these differences in molecular geometry. You can put sulfur in the middle because fluorine tends to make single bonds. 
The central atom is usually the least electronegative element in the compound, and in KrF4, Krypton fits this criterion. In this case, the central atom is Krypton (Kr). Within the context of VSEPR theory, you can count electrons to determine the electron geometry ("parent" geometry). To begin drawing the Lewis structure of KrF4 (Krypton Tetrafluoride), we first need to identify the central atom. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
